https://iupac.qmul.ac.uk/AtWt/table.html
World Wide Web version prepared by G. P. Moss
School of Biological and Chemical Sciences, Queen Mary University of London,
Mile End Road, London,
E1 4NS, UK
g.p.moss@qmul.ac.uk
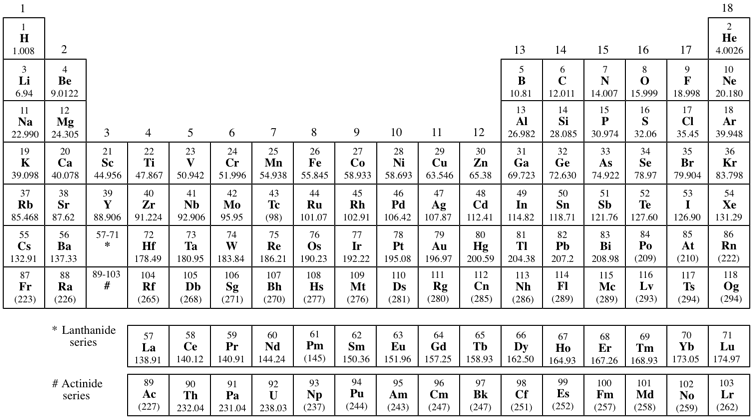
This version of the Periodic table is based on that recommended by the International Union of Pure and Applied Chemistry and published in IUPAC Nomenclature of Inorganic Chemistry, Recommendations 2005. Atomic weights are quoted to five significant figures. For more precise values see the table of 2017 recommended values and for the 2017 changes see IUPAC website. For elements with no stable nuclides the mass of the longest-lived isotope has been quoted in brackets. However three such elements (Th, Pa, and U) do have a characteristic terrestrial isotopic composition, and for these an atomic weight is tabulated. The names for elements above 118 may be referred to by temporary names based on the 1978 recommendations (see Pure Appl. Chem., 1979, 51, 381-384). The discovery of elements 113, 115, 117 and 118 have been verified by IUPAC. See Pure Appl. Chem., 2016, 88, 139-153 for elements 113, 115 and 117 and Pure Appl. Chem., 2016, 88, 155-160 for element 118. They have been named nihonium (Nh), moscovium (Mc), tennessine (Ts) and oganesson (Og) Pure Appl. Chem., 2016, 88, 1225-1229.

Return to IUPAC Chemical Nomenclature home page