Nomenclature of Carbohydrates (Recommendations 1996)
2-Carb-13
Continued from 2-Carb-11 and 2-Carb-12
Contents
2-Carb-13. Deoxy sugars
2-Carb-13.1. Trivial names
Several deoxy sugars have trivial names established by long usage, e.g. fucose (Fuc), quinovose (Qui) and rhamnose (Rha). They are illustrated here in the pyranose form. These names are retained for the unmodified sugars, but systematic names are usually preferred for the formation of names of derivatives, especially where deoxygenation is at a chiral centre of the parent sugar. (See also the alphabetical listing of trivial names in the Appendix.)
Examples:
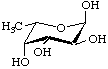
α-L-Fucopyranose
6-Deoxy-α-L-galactopyranose
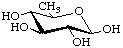
β-D-Quinovopyranose
6-Deoxy-β-D-glucopyranose
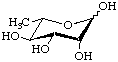
L-Rhamnopyranose
6-Deoxy-L-mannopyranose
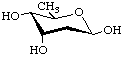
2,6-Dideoxy-β-D-ribo-hexopyranose
(β-Digitoxopyranose)

3,6-Dideoxy-β-D-xylo-hexopyranose
(β-Abequopyranose)
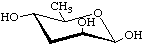
3,6-Dideoxy-β-D-arabino-hexopyranose
(β-Tyvelopyranose)
Other trivial names that have been used include ascarylose for 3,6-dideoxy-L-arabino-hexose, colitose for 3,6-dideoxy-L-xylo-hexose and paratose for 3,6-dideoxy-D-ribo-hexose.
Note. Sugars with a terminal CH3 group should be named as ω-deoxy sugars, as shown above, not C-methyl derivatives.
2-Carb-13.2. Names derived from trivial names of sugars
Use of 'deoxy-' in combination with an established trivial name (see Charts I and II) is straightforward if the formal deoxygenation does not affect the configuration at any asymmetric centre. However if 'deoxy' removes a centre of chirality, the resulting names contain stereochemical redundancy. In such cases, systematic names are preferred, especially for the naming of derivatives.
Note. The names 2-deoxyribose (for 2-deoxy-D-erythro-pentose) and 2-deoxyglucose (for 2-deoxy-D-arabino-hexose) are often used.
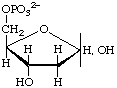
2-Deoxy-D-erythro-pentofuranose 5-phosphate
2-Carb-13.3. Systematic names
The systematic name consists of the prefix 'deoxy-', preceded by the locant and followed by the stem name with such configurational prefixes as necessary to describe the configuration(s) at the asymmetric centres present in the deoxy compound. Configurational prefixes are cited in order commencing at the end farthest from C-1. 'Deoxy' is regarded as a detachable prefix, i.e. it is placed in alphabetical order with any substituent prefixes.
Note. The treatment of 'anhydro' (see 2-Carb-26), 'dehydro' (see 2-Carb-17.3) and 'deoxy' as detachable prefixes follows long-standing practice in carbohydrate chemistry, but is in conflict with [14] (p. 12).
Examples:
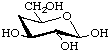
4-Deoxy-β-D-xylo-hexopyranose not 4-deoxy-β-D-galactopyranose
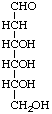
2-Deoxy-D-ribo-hexose not 2-deoxy-D-allose
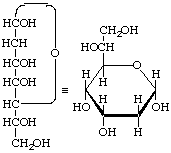
2-Deoxy-α-D-allo-heptopyranose
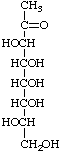
1-Deoxy-L-glycero-D-altro-oct-2-ulose
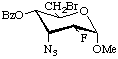
Methyl 3-azido-4-O-benzoyl-6-bromo-2,3,6-trideoxy-2-fluoro-α-D-allopyranoside
If the CH2 group divides the chiral centres into two sets, it is ignored for the purpose of assigning a configurational prefix; the prefix(es) assigned should cover the entire sequence of chiral centres (see 2-Carb-8.4).
Examples:
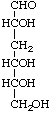
3-Deoxy-D-ribo-hexose not 3-deoxy-D-erythro-D-glycero-hexose
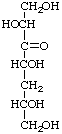
5-Deoxy-D-arabino-hept-3-ulose not 5-deoxy-D-glycero-D-glycero-L-glycero-hept-3-ulose
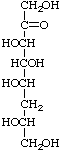
6-Deoxy-L-gluco-oct-2-ulose not 6-deoxy-L-glycero-L-xylo-oct-2-ulose
If the anomeric hydroxy group is replaced by a hydrogen atom, the compound is named as an anhydro alditol (2-Carb-26).
2-Carb-13.4. Deoxy alditols
The name of an aldose derivative in which the aldehyde group has been replaced by a terminal CH3 group is derived from that of the appropriate alditol (see 2-Carb-19) by use of the prefix 'deoxy-'.
Examples:
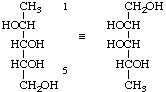
1-Deoxy-D-arabinitol not 5-deoxy-D-lyxitol
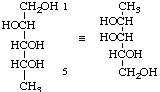
5-Deoxy-D-arabinitol not 1-deoxy-D-lyxitol
The alditols from fucose and rhamnose are frequently termed fucitol and rhamnitol (see 2-Carb-19.1).
Reference
14. Guide to IUPAC Nomenclature of Organic Compounds, Recommendations 1993, Blackwell Scientific Publications, Oxford (1993).
Continue to the next section with 2-Carb-14 and 2-Carb-15 of Nomenclature of Carbohydrates.
Return to Carbohydrates home page.