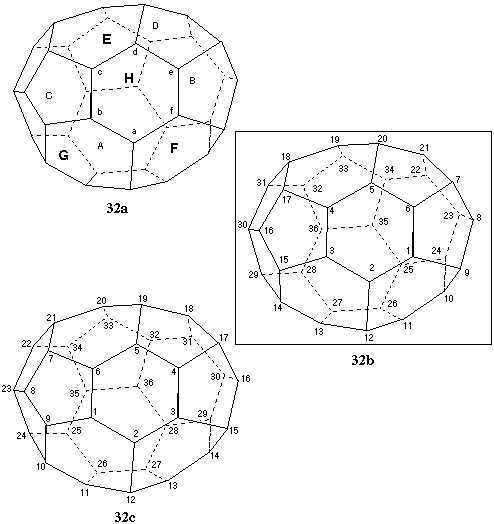
Fig. 32. Systematic numbering of (f,sC)(C36-C1)[5,6]fullerene
This fullerene contains eight six-membered rings indicated by the letters A to H in Fig. 32a (smaller-font letters denote rings away from the viewer; larger-font, bold letters denote rings closer to the viewer). According to Fu-3.4.1, ring H is preferred to begin the numbering since it contains the highest-ranking atom at the first point of difference. There are twelve pathways to be considered in ring H, clockwise and anticlockwise from each atom a - f. In order to reduce the number of pathways to be actually examined, those beginning from the 6,6,6 atom (clockwise and anticlockwise) are studied first, because, if contiguous, these pathways will be preferred to any other pathway starting in this ring according to Fu-3.4.2. Unfortunately, they are both discontiguous. The two pathways having the 6,6,6 atom a at position 2 are then examined for contiguous numbering: again these would be preferred at this point, since no other pathway can have a 6,6,6 atom at position 2. The two pathways are clockwise from atom f, and anticlockwise from atom b. They lead to the contiguous numberings shown in Figs. 32b and 32c, respectively. These contiguous sequences are then compared using Fu-3.4.2 to discover that there is a 6,6,5 atom at position 4 in the pathway from atom f (Fig. 32b) and a 6,5,5 atom at the same position in the pathway from atom b (Fig. 32c). Therefore the numbering sequence shown in Fig. 32b, is correct. The enantiomer shown here has the (f,sC) [ref 4].

Fig. 32. Systematic numbering of (f,sC)(C36-C1)[5,6]fullerene
4. International Union of Pure and Applied Chemistry, Division of Organic Chemistry, Commission on Nomenclature of Organic Chemistry. "Nomenclature for the (C60-Ih) and (C70-D5h(6))[5,6]fullerenes (IUPAC recommendations 2002)", Pure Appl. Chem. 2002, 74, 629-695.
5. P.W. Fowler and D.E. Manolopoulos, An Atlas of Fullerenes, Clarendon Press, Oxford, 1995