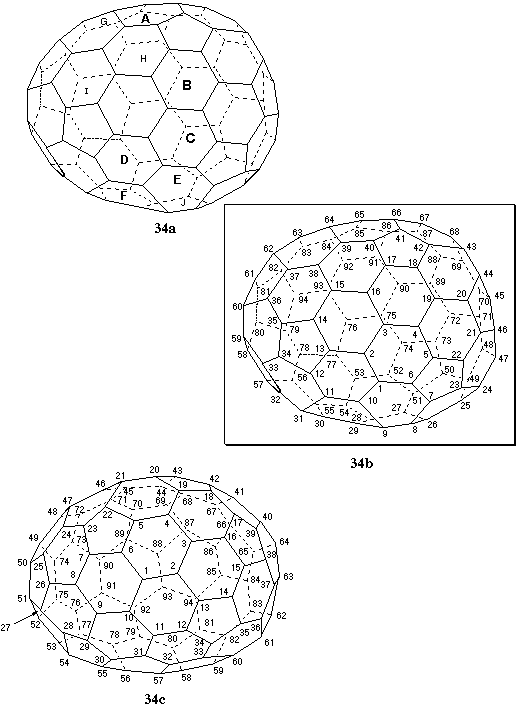
Fig. 34. Systematic numbering of (f,sC)(C94-C1)[5,6]fullerene
This fullerene contains only 6,6,6 or 6,6,5 atoms and thirty-six six-membered rings. Seventeen rings contain a sequence of four 6,6,6 and two 6,6,5 atoms and the numbering should begin from these rings according to Fu-3.4.1. Of all the possible numberings, the seventeen clockwise and the seventeen anticlockwise pathways featuring the four 6,6,6 atoms in a row at the beginning of the numbering are considered first, since, if contiguous, they will be preferred according to rule Fu-3.4.2. Eleven of these pathways lead to contiguous numberings: clockwise from rings A, B, C, E, and F; anticlockwise from rings D, E, G, H, I, and J (Fig. 34a; smaller-font letters denote rings away from the viewer; larger-font, bold letters denote rings closer to the viewer). Comparison of the atom sequences by application of Fu-3.4.2 eliminates five numberings because of the ranking of the atoms at position 8, two of the six remaining numberings at position 9, and two of the four remaining numberings at position 11. This leaves only the two numberings reported in Figs. 34b (clockwise from ring C) and 34c (anticlockwise from ring J). Selection between these can be made by inspection of the atoms up to position 15, which is a 6,6,6 atom in the numbering of Fig. 34b and a 6,6,5 atom in the numbering of Fig. 34c. The former is therefore preferred based on Fu-3.4.2. The configuration of the shown enantiomer is (f,sC) [ref 4].

Fig. 34. Systematic numbering of (f,sC)(C94-C1)[5,6]fullerene
Finally, it must be noted that this document does not deal with Cs, Ci, and C1 fullerenes without contiguous numbering, of which no examples were encountered during the elaboration of the present document. Since these fullerenes have a large number of possible numbering pathways, it seems rather unlikely that a contiguous numbering cannot be found for most of these structures. In any case, application of Rules Fu-3.2.1.a, Fu-3.2.3, and Fu-3.2.4 should allow the user to identify a preferred discontiguous numbering.
1. International Union of Pure and Applied Chemistry. "Nomenclature and Terminology of Fullerenes: A Preliminary Report", E.W. Godly and R. Taylor, Pure Appl. Chem. 1997, 69, 1411-1434.
2. R. Taylor, J. Chem. Soc., Perkin Trans. 2 1993, 813-824.
3. A.L. Goodson, C.L. Gladys and D.E. Worst, J. Chem. Inf. Comp. Sci. 1995, 35, 969-978.
4. International Union of Pure and Applied Chemistry, Division of Organic Chemistry, Commission on Nomenclature of Organic Chemistry. "Nomenclature for the (C60-Ih) and (C70-D5h(6))[5,6]fullerenes (IUPAC recommendations 2002)", Pure Appl. Chem. 2002, 74, 629-695.
5. P.W. Fowler and D.E. Manolopoulos, An Atlas of Fullerenes, Clarendon Press, Oxford, 1995