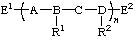 | regular single-strand polymer |
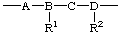 | constitutional repeating unit (CRU) |
| -A-, -B-, -C-, -D- | subunits |
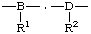 | substituted subunits |
| R1, R2 | substituents to subunits |
| E1, E2 | end-groups |
In 1952, the Subcommission on Nomenclature of the IUPAC Commission on Macromolecules published a report [ref 1] on the nomenclature of polymers that included a method for the systematic naming of linear organic polymers on the basis of structure. A later report [ref 2] dealing with steric regularity utilized that system of nomenclature. When the first report was issued, the skeletal rules were adequate for most needs; indeed, most polymers could at that time be reasonably named on the basis of the real or hypothetical substance used in producing the polymer. In the intervening years, however, the rapid growth of the polymer field had dictated a need for modification and expansion of the earlier rules. The result was a set of rules approved in 1975 [ref 3]. The present report is an updating of the 1975 rules with special attention to developments in the nomenclature of organic chemistry since that time [ref 4, ref 5].
The rules in the present report are designed to name, uniquely and unambiguously, the structures of regular single-strand organic polymers whose repeating structures can be written within the framework of ordinary chemical principles. Although the stereochemistry of polymers is not considered here, examples of names with stereodescriptors are given. A detailed survey of stereochemical notation of polymers is given in a special report [ref 6]. As with organic chemistry nomenclature, this nomenclature describes chemical structures rather than substances. It is realized that a polymeric substance ordinarily may include many structures, and that a complete description of even a single polymer molecule would include an itemization of terminal groups, branching, random impurities, degree of steric regularity, chain imperfections, etc.
Nevertheless, it is useful to think of the macromolecules of a polymer as being represented by a single structure that may itself be hypothetical. To the extent that the structure can be portrayed as a chain of regular structural repeating units (SRUs) or constitutional repeating units (CRUs) (the terms are synonymous), the structure can be named by the rules in this report; in addition, provision has been made for including end-groups in the name.
The fundamental principles and the basic rules of the structure-based nomenclature are given first, accompanied by detailed extensions and applications. An Appendix contains names of common sub-units as well as a list of acceptable source-based names, along with the corresponding structure-based names, of common polymers. The Commission sees no objection to the continued use of such source-based names where these names are clear and unambiguous, but prefers the use of the structure-based nomenclature detailed in these rules.
The rules of structure-based nomenclature of regular single-strand polymers are of fundamental importance in polymer nomenclature. The names of other kinds of polymers such as double-strand [ref 7] and irregular polymers [ref 8] are based on the principles given in this Report.
After more than two decades of use, many improvements in the 1975 rules [ref 3] have been suggested. As a result, the present new rules are proposed. The new rules do not represent any change in principles. They involve mainly rearrangement of the material, a generalization of basic rules, a clearer presentation, an avoidance of manifold repetition of the same principles at various places, and the use of graphical means for the visualization of the principles. Also, some additions are made, such as a rule on naming polymer chain substituents, and several new examples of polymers, including those with an "inorganic" backbone and those where stereodescriptors are essential.
Regular polymer [ref 9]
A polymer composed of regular macromolecules, i.e., macromolecules the structure of which essentially comprises the repetition of a single constitutional unit with all units connected identically with respect to directional sense.
Single-strand polymer [ref 3, ref 7, ref 9, ref 10]
A polymer, the macromolecules of which are single-strand macromolecules, i.e., macromolecules comprising constitutional units connected in such a way that adjacent constitutional units are joined to each other through two atoms, one on each constitutional unit.
Constitutional unit [ref 9]
An atom or group of atoms (with pendant atoms or groups, if any) comprising a part of the essential structure of a macromolecule, an oligomer molecule, a block, or a chain.
Constitutional repeating unit (CRU) [ref 9]
The smallest constitutional unit, the repetition of which constitutes a regular macromolecule, a regular oligomer molecule, a regular block, or a regular chain.
Main chain (backbone) [ref 9]
That linear chain to which all other chains, long or short or both, may be regarded as being pendant.
End-group [ref 9]
A constitutional unit that is an extremity of a macromolecule or oligomer molecule.
Subunit
The largest main-chain (backbone) segment of the CRU that can be named as a single unit under organic nomenclature rules [ref 4, ref 5]. This may be a ring or ring system, a heteroatom or a homogeneous heteroatom chain, or an acyclic carbon chain.
Path length
The path length between two subunits is the number of polymer main-chain (backbone) atoms between the two subunits. Where a ring or ring system constitutes all or part of a path between two subunits, the shortest continuous chain of atoms in the ring or ring system is selected.
Seniority
Priority in a set of atoms or groups of atoms according to a prescribed order.
Locant
A numeral or letter that identifies position in a structure.
3. Fundamental Principles
regular single-strand polymer constitutional repeating unit (CRU) -A-, -B-, -C-, -D- subunits substituted subunits R1, R2 substituents to subunits E1, E2 end-groups
This nomenclature method rests upon the selection of a preferred CRU of which the polymer molecule is a multiple. Wherever possible, the CRU and subunits are named according to the IUPAC-recommended nomenclature of organic chemistry [ref 4, ref 5].
In this nomenclature, the steps to be followed when naming a polymer are:
| 1. | Write the structure of the polymer chain. A sufficient portion of the chain should be written to show structure repetition. The portion that repeats is a CRU. |
| 2. | Select the preferred CRU. |
| 3. | Name the preferred CRU by citing, from left to right, the names of the subunits, including their substituents, if present. |
| 4. | Name the polymer. |
Structure of the polymer chain
In simple cases, the CRU involves a single subunit. In more complex cases, it is often necessary to draw a large segment of the polymer chain, for example,
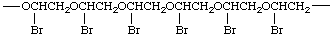
There are many ways to write the CRU for most chain structures. In simple cases, these units are readily identified. In the polymer chain given above, the possible CRUs are
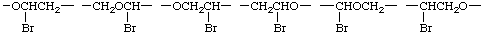
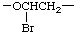 or
or 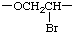
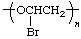 | rather than | 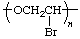 |
| 1-substitution | 2-substitution |
The name of the preferred CRU is formed by citing, in the order in which they appear in the CRU, the names of the subunits within the CRU. In the example, the oxygen atom is called oxy and the -CH2CH2- (preferred to -CH2- because it is larger and can be named as a unit) is called ethylene; the latter unit substituted with one bromine atom is called 1-bromoethylene. The preferred CRU is, therefore, named oxy(1-bromoethylene).
Naming the polymer
The name of the polymer is simply the name of the preferred CRU enclosed in curves, square brackets, or braces and prefixed by poly. The nesting order of enclosing marks is curves, square brackets, braces, then curves, square brackets, braces, etc., i.e., {[({[( )]})]}. This is well illustrated in Examples 21 and 31 in Section 9.
The polymer is named poly[oxy(1-bromoethylene)].
1. IUPAC. "Report on nomenclature in the field of macromolecules", J. Polym. Sci. 8, 257-277 (1952).
2. IUPAC. "Report on nomenclature dealing with steric regularity in high polymers", Pure Appl. Chem. 12, 643-656 (1966).
3. IUPAC. "Nomenclature of regular single-strand organic polymers 1975", Pure Appl. Chem. 48, 373-385 (1976). Reprinted as Chapter 5 in IUPAC. Compendium of Macromolecular Nomenclature, Blackwell Scientific Publications, Oxford (1991) (PDF).
4. IUPAC. Nomenclature of Organic Chemistry, Sections A, B, C, D, E, F and H, Pergamon Press, Oxford (1979).
5. IUPAC. A Guide to IUPAC Nomenclature of Organic Compounds, Blackwell Scientific Publications, Oxford (1993).
6. IUPAC. "Stereochemical definitions and notations relating to polymers 1980", Pure Appl. Chem. 53, 733-752 (1981). Reprinted as Chapter 2 in IUPAC. Compendium of Macromolecular Nomenclature, Blackwell Scientific Publications, Oxford (1991) (PDF).
7. IUPAC. "Nomenclature of regular double-strand (ladder and spiro) organic polymers 1993", Pure Appl.Chem. 65, 1561-1580 (1993).
8. IUPAC. "Structure-based nomenclature for irregular single-strand organic polymers 1993", Pure Appl.Chem. 66, 873-889 (1994).
9. IUPAC. "Glossary of basic terms in polymer science 1996", Pure Appl.Chem. 68, 2287-2311 (1996).
10. IUPAC. "Nomenclature for regular single-strand and quasi-single-strand inorganic and coordination polymers 1984", Pure Appl.Chem. 57, 149-168 (1985).Reprinted as Chapter 6 in IUPAC. Compendium of Macromolecular Nomenclature, Blackwell Scientific Publications, Oxford (1991) (PDF) and as Chapter II-7 in IUPAC. Nomenclature of Inorganic Chemistry II, Royal Society of Chemistry, Cambridge (2001).