| 1. | CF3-CH2[2H] | 1,1,1-Trifluoro[2H1]ethane |
| 2. | 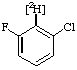 | 1-Chloro-3-fluoro[2-2H]benzene |
| 3. | 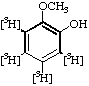 | 2-Methoxy[3,4,5,6-3H4]phenol |
Continued from H-2, Names for Isotopically Modified Compounds
CONTENTS
Rule H-3.1. Numbering in relation to the unmodified compound
3.11 - Numbering of an isotopically modified compound is not changed from that of an isotopically unmodified compound. Among the structural features of a compound to be considered successively for numbering as given by the Rule C-15.11 and Section E (ref. 6), the presence of nuclides is considered last with the exception of chirality arising from isotopic modification (see Rule H-3.22, example 7).
Examples:
| 1. | CF3-CH2[2H] | 1,1,1-Trifluoro[2H1]ethane |
| 2. |  | 1-Chloro-3-fluoro[2-2H]benzene |
| 3. |  | 2-Methoxy[3,4,5,6-3H4]phenol |
Note:
One should be aware that, when isotopically modified compounds are named by the system based on an extension of the Boughton principles (see Introduction), lowest locants are assigned to isotopic positions included in the parent structure, which includes unsaturation and principal groups, if any, before other considerations. This sometimes results in the assignment of higher locants for substituents expressed by prefixes, which may give a numbering that differs from the one assigned according to Rule H-3.1.
Example:
| F3C-CH22H | 2,2,2-Trifluoroethane-1-d |
Note that here the presence of deuterium (d) causes the carbon atom to which it is attached to be assigned the locant 1. For more details and examples see ref. 4.
Rule H-3.2. Priority between isotopically modified and unmodified atoms or groups
3.21 - When there is a choice between equivalent possibilities for the principal chain or senior cyclic system in an isotopically unmodified compound, the principal chain or senior cyclic system of the analogous isotopically modified compound is chosen so as to include the maximum number of modified atoms or groups. If a choice still remains, precedence is given to the principal chain or senior ring that contains a nuclide of higher atomic number. In case of different nuclides of the same element, precedence is given to a nuclide of higher mass number.
Examples:
| 1. | 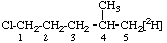 | 1-Chloro-4-methyl[5-2H1]pentane not 1-Chloro-4-([2H1]methyl)pentane |
| 2. | 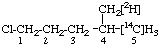 | 1-Chloro-4-([2H1]methyl)[5-14C]pentane not 1-Chloro-4-([14C]methyl)[5-2H1]pentane |
| 3. | 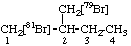 | 1-([81Br]Bromo)-2-([79Br]bromomethyl)butane not 1-([79Br]Bromo)-2-([81Br]bromomethyl)butane |
3.22 - When there is a choice between equivalent numberings in an isotopically unmodified compound, the starting point and direction of numbering of the analogous isotopically modified compound are chosen so as to give lowest locants to the modified atoms or groups considered together in one series in ascending numerical order (see Rule C-15.11, ref. 6). If a choice still remains, precedence for the lowest locants is given to a nuclide of higher atomic number. In the case of different nuclides of the same element, precedence is given to the nuclide of higher mass number.
Examples:
| 1. | 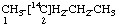 | [2-14C]Butane not [3-14C]Butane |
| 2. | 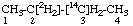 | [3-14C,2,2-2H2]Butane not [2-14C,3,3-2H2]Butane |
| 3. | 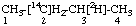 | [2-14C,3-2H1]Butane not [3-14C,2-2H1]Butane |
| 4. | 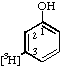 | [3-3H]Phenol |
| 5. | 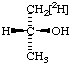 | (R)-[1-2H1]-2-Propanol |
| 6. | 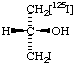 | (S)-1,3-[1-125I]Diiodo-2-propanol |
| 7. | 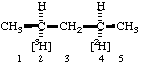 | (2S,4R)-[4-2H1,2-3H1]Pentane not (2R,4S)-[2-2H1,4-3H1]Pentane |
Note:
In the last example above, the numbering follows Rule H-3.22 rather than the stereochemical preferences as described in Rules of Section E, (ref. 6) which give preference to R groups over S groups for lowest locants.
H-4. LOCANTS FOR NUCLIDES IN ISOTOPICALLY MODIFIED COMPOUNDS
Rule H-4.1. Omission or introduction of locants
4.11 - When there is no ambiguity, locants may be omitted from the isotopic designator in the name of an isotopically modified compound.
Examples:
| 1. | C[2H3]-CN | [2H3]Acetonitrile |
| 2. | CH3-NH[2H] | Methyl[2H1]amine |
| 3. | CH3-CH2-O[2H] | Ethan[2H]ol |
| 4. | [2H]O-CH2-CH2-O[2H] | 1,2-Ethane[2H2]diol |
| 5. | CH2[2H]-O-C[2H2]-S-CH2-OOH | [([2H1]Methoxy[2H2]methyl)thio]methyl hydroperoxide |
4.12 - When ambiguity may occur, specific positions of nuclides should be indicated in the isotopic designator by appropriate locants, letters and/or numerals, preceding the nuclide symbol(s).
Examples (see also Rules H-2.11, H-2.21, H-2.31):
4.21 - When a nuclide occupies a position that is not numbered, an italicized prefix or Greek letter may be used to denote its position.
Examples:
4.22 - When a nuclide occupies a position that is not numbered or when its position cannot be easily defined according to Rules H-4.12 and H-4.21, the nuclide symbol may be included in the entire symbol of the group through which it is linked to the main part of the structure.
Examples:
4.23 - Italicized nuclide symbols and/or capital italic letters may be used as locants to distinguish between different nuclides of the same element.
Examples:
1. I.U.P.A.C. Manual of Symbols and Terminology for Physicochemical Quantities and Units, 1973 Edition, Butterworths, London, 1975, Rules 7.1 and 7.2, p. 24.
2. I.U.P.A.C. Commission on Atomic Weights, Pure and Applied Chemistry, 37, 59l-603 (1974).
3. (a) W. A. Boughton, Naming Hydrogen Isotopes, Science, 79, 159-60 (1934); (b) E. J. Crane, Nomenclature of the Hydrogen Isotopes and Their Compounds, Science, 80, 86-9 (1934); (c) American Chemical Society, Report of Committee on Nomenclature, Spelling, and Pronunciation, Nomenclature of the Hydrogen isotopes and Their Compounds, Ind. Eng. Chem. (News Ed.), 13, 200-1 (1935).
4. "The Naming and Indexing of Chemical Substances for Chemical Abstracts During the Ninth Collective Period (1972-1976)", ¶ 220, p. 111I, a reprint of Section IV of the Introduction to the Chemical Abstracts Volume 76 Index Guide.
5. I.U.P.A.C. Nomenclature of Inorganic Chemistry, 2nd Edition (1970), Butterworths, London. 1971: (a) Rule 1.31, p. 11; (b) Rule 1.1, p. 10.
6. I.U.P.A.C. Nomenclature of Organic Chemistry, Sections A, B, C, D, E, F and H (1979 edition), Pergamon Press, Oxford. 1979.
Comparative Examples of Formulae and Names for Isotopically Modified Compounds
1. C[2H3]-NH2 [1,1,1-2H3]Methylamine 2. CH3-CH[2H]-OH [1-2H1]Ethanol 3. HO-CH[2H]-CH[2H]-OH [1,2-2H2]-1,2-Ethanediol 4. 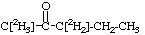
[1,1,1,3,3-2H5]-2-Pentanone 5. 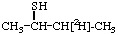
[3-2H1]-2-Butanethiol 6. 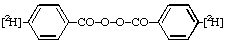
Di([4-2H]benzoyl) peroxide 7. 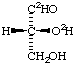
(R)-(O2,1-2H2)Glyceraldehyde
or
(R)-(O-2H)Glycer(2H)aldehyde8. 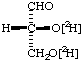
(R)-[O2,O3-2H2]Glyceraldehyde 9. [2H]S-CH2-CH(N[2H2])-CO2[2H] DL-[N,N,O,S-2H4]Cysteine
1. [2H3][14C]-S-CH2-CH2-CH(NH2)-COOH L-[methyl-(14C,2H3)]Methionine 2. 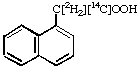
1-Naphthalene[carboxy-14C, ,
-2H2]acetic acid
3. 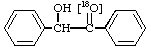
[carbonyl-18O]Benzoin 4. 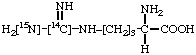
L-[guanidino-14C,N3-15N]Arginine
1. CH3-CH2-S-[34S]-S-(CH2)2-CO2H 3-(Ethyl[S-34S-S]trithio)propionic acid
or
3-(Ethyl[2-34S]trisulfanyl)propionic acid
(see Rule C-515.2, ref 6)2. CH3-CH2-[34S]-S-S-(CH2)2-CO2H 3-(Ethyl[34S-S-S]trithio)propionic acid
or
3-(Ethyl[3-34S]trisulfanyl)propionic acid
(see Rule C-515.2, ref 6)3. 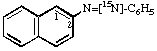
Naphthalene-2-[N=15N]azobenzene 4. 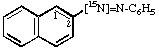
Naphthalene-2-[15N=N]azobenzene
1. 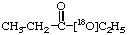
18O-Ethyl [18O1]propionate 2. 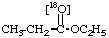
O-Ethyl [18O1]propionate 3. 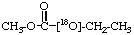
18O-Ethyl O-methyl [18O1]carbonate 4. 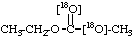
O-Ethyl 18O-methyl [18O2]carbonate 5. 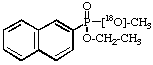
O-Ethyl 18O-methyl 2-naphthyl[18O1]phosphonate 6. 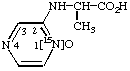
N-([1-15N]-2-Pyrazinyl)alanine 15N-oxide
Type of Compound Formula Name Unmodified CH3-CH2-OH Ethanol Isotopically substituted C2H3-CH2-O2H (2,2,2-2H3)Ethan(2H)ol
or
(O,2,2,2-2H4)EthanolSpecifically labeled C[2H3]-CH2-O[2H] [2,2,2-2H3]Ethan[2H]ol
or
[O,2,2,2-2H4]EthanolSelectively labeled a. [O,2-2H]CH3-CH2-OH
b. [2-2H2;218O0;l]CH3-CH2-OHa. [O,2-2H]Ethanol
b. [2-2H2;2,18O0;1]EthanolNonselectively labeled [2H]CH3-CH2-OH [2H]Ethanol Isotopically deficient [def13C]CH3-CH2-OH [def13C]Ethanol
Return to Section H: Isotopic Modified Compounds home page.