Extension and Revision of the von Baeyer System for Naming Polycyclic Compounds (Including Bicyclic Compounds)
(IUPAC Recommendations 1999)
VB-8.1 and VB-8.2
Continued from VB-7
Contents of Section
VB-8 Naming Modified Ring Systems (Heteroatoms, unsaturation, stereochemistry, etc.)
VB-8.1 Heteroatoms
VB-8.2 Principal Functional Group
References for this Section
Continued with VB-8.3 Unsaturation
VB-8 Naming Modified Ring Systems (Heteroatoms, unsaturation, stereochemistry, etc.)
Heteroatoms are indicated by replacement terms (see B-4.2, B-6.1, R-1.2.2.1, R-9.3); unsaturation is indicated in the usual way by the ending -ene, -diene, etc. (see A-11.3, R-3.1.1); stereochemistry by the R/S notation (see E-4.9, R-7.2.1); and substituents and the principal functional group in the usual way (refs 6, 7).
If there is still a choice of name or numbering the following criteria are considered in order until a decision is made:
VB-8.1 Low locants are allocated for heteroatoms indicated by replacement terms considered together as a set in ascending numerical order.
Examples
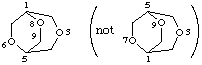
3,6,8-trioxabicyclo[3.2.2]nonane
not 3,7,9-trioxabicyclo[3.2.2]nonane (3,6,8 is lower than 3,7,9)
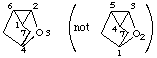
However 3-oxatricyclo[2.2.1.02,6]heptane
not 2-oxatricyclo[2.2.1.03,5]heptane (the bridge locants determine the numbering; 2,6 is lower than 3,5; see VB-6.4)
VB-8.1.1 Low locants are allocated for heteroatoms in the order of RB-1.4 (ref 13), R-2.3.3 and R-9.3 (ref 7).
Example
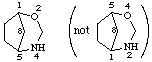
2-oxa-4-azabicyclo[3.2.1]octane
not 4-oxa-2-azabicyclo[3.2.1]octane (2-oxa is lower than 4-oxa)
VB-8.1.2 Low locants are allocated for non-standard valency expressed by the ![[lambda]](../greek/LAMBDA.GIF) symbol (ref 8), if necessary in order of decreasing numerical value of the bonding number, i.e. lower numbers for
symbol (ref 8), if necessary in order of decreasing numerical value of the bonding number, i.e. lower numbers for ![[lambda]](../greek/LAMBDA.GIF) 6 rather than
6 rather than ![[lambda]](../greek/LAMBDA.GIF) 4.
4.
Examples
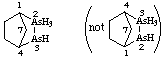
2![[lambda]](../greek/LAMBDA.GIF) 5,3-diarsabicyclo[2.2.1]heptane
5,3-diarsabicyclo[2.2.1]heptane
not 2,3![[lambda]](../greek/LAMBDA.GIF) 5-diarsabicyclo[2.2.1]heptane (2 is lower than 3)
5-diarsabicyclo[2.2.1]heptane (2 is lower than 3)
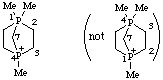
1,1,4-trimethyl-1![[lambda]](../greek/LAMBDA.GIF) 5,4-diphosphabicyclo[2.2.1]heptan-4-ium
5,4-diphosphabicyclo[2.2.1]heptan-4-ium
not 1,4,4-trimethyl-1,4![[lambda]](../greek/LAMBDA.GIF) 5-diphosphabicyclo[2.2.1]heptan-1-ium (1
5-diphosphabicyclo[2.2.1]heptan-1-ium (1![[lambda]](../greek/LAMBDA.GIF) 5 is lower than 4
5 is lower than 4![[lambda]](../greek/LAMBDA.GIF) 5)
5)
VB-8.1.3 Low locants are allocated for radical sites, or if the ring system is a substituent, its point of attachment.
Examples
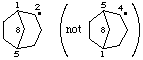
bicyclo[3.2.1]octan-2-yl
not bicyclo[3.2.1]octan-4-yl (2 is lower than 4)
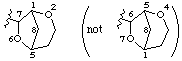
However 2,6-dioxabicyclo[3.2.1]octan-7-yl
not 4,7-dioxabicyclo[3.2.1]octan-6-yl (2,6 is lower than 4,7, see VB-8.1)
Note In previous editions of these rules (A-31.4 and B-14.1, ref 6) octan-yl was contracted to oct-yl [however see R-2.5 and R-5.8.1 (ref 7) and RC-81.1.2 (ref 9) where the contraction is not used].
VB-8.1.4 Low locants are allocated for cationic sites.
Examples
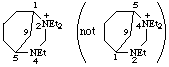
2,2,4-triethyl-2,4-diazabicyclo[3.3.1]nonan-2-ium (preferred name)
Alternative name 2,2,4-triethyl-4-aza-2-azoniabicyclo[3.3.1]nonane
not 2,4,4-triethyl-2,4-diazabicyclo[3.3.1]nonan-4-ium (2-ium is lower than 4-ium)

1![[lambda]](../greek/LAMBDA.GIF) 4,3,5
4,3,5![[lambda]](../greek/LAMBDA.GIF) 4,7-tetrathiabicyclo[3.3.1]nonan-1-ylium (preferred name)
4,7-tetrathiabicyclo[3.3.1]nonan-1-ylium (preferred name)
Alternative name 3,5![[lambda]](../greek/LAMBDA.GIF) 4,7-trithia-1-thioniabicyclo[3.3.1]nonane
4,7-trithia-1-thioniabicyclo[3.3.1]nonane
not 1![[lambda]](../greek/LAMBDA.GIF) 4,3,5
4,3,5![[lambda]](../greek/LAMBDA.GIF) 4,7-tetrathiabicyclo[3.3.1]nonan-5-ylium (1-ylium is lower than 5-ylium)
4,7-tetrathiabicyclo[3.3.1]nonan-5-ylium (1-ylium is lower than 5-ylium)
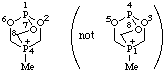
4-methyl-2,6,7-trioxa-1,4-diphosphabicyclo[2.2.2]octan-4-ium (preferred name)
Alternative name 4-methyl-2,6,7-trioxa-1-phospha-4-phosphoniabicyclo[2.2.2]octane
not 1-methyl-3,5,8-trioxa-1,4-diphosphabicyclo[2.2.2]octan-1-ium (1,2,4,6,7 is lower than 1,3,4,5,8; see VB-8.1)
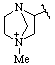
However 4-methyl-1,4-diazabicyclo[2.2.1]heptan-4-ium-2-yl
not 1-methyl-1,4-diazabicyclo[2.2.1]heptan-1-ium-3-yl (2 is lower than 3 for free valence; see VB-8.1.3)
VB-8.2 Low locants are allocated for the principal functional groups.
Examples
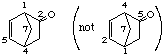
bicyclo[2.2.1]hept-5-en-2-one
not bicyclo[2.2.1]hept-2-en-5-one (2-one is lower than 5-one)
References for this Section
6. International Union of Pure and Applied Chemistry, Nomenclature of Organic Chemistry, Sections A, B, C, D, E, F and H, 1979 edition, Pergamon Press, Oxford, 1979.
7. International Union of Pure and Applied Chemistry, A Guide to IUPAC Nomenclature of Organic Compounds, Recommendations 1993, Blackwell Scientific Publications, Oxford, 1993.
8. IUPAC Commission on the Nomenclature of Organic Chemistry, Treatment of variable valence in organic nomenclature (Lambda convention) (Recommendations 1983), Pure Appl. Chem., 56, 769-778 (1984).
9. IUPAC Commission on the Nomenclature of Organic Chemistry, Revised nomenclature for radicals, ions, radical ions and related species (IUPAC recommendations 1993), Pure Appl. Chem., 65, 1357-1455 (1993).
13. IUPAC Commission on the Nomenclature of Organic Chemistry, Revision of the extended Hantzsch-Widman system of nomenclature for heteromonocycles (Recommendations 1982), Pure Appl. Chem., 55, 409-416 (1983).
Continued with VB-8.3 to VB-8.5
Return to von Baeyer Nomenclature home page.