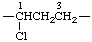 |  |
| 1-chloropropane-1,3-diyl | cyclohexane-1,4-diyl |
Contents of this section
The CRU in a polymer chain may contain one or more subunits. A simple CRU is one that can be described by single subunit. A complex CRU has at least two subunits.
In selecting the preferred CRU, the steps to be followed in sequence are:
| 1. | write the structure of a representative portion of the polymer chain |
| 2. | identify the single subunit and its substituents; |
| 3. | choose the direction so that the locants of free valences of the subunit are as low as possible; |
| 4. | choose the direction so that the locants of the substituents are as low as possible. |
For acyclic and monocarbocyclic subunits, preference in lowest numbers is given to the carbon atoms through which they are attached to the main chain of the CRU. The point of attachment at the left-hand side of the subunit as written in the CRU must have locant 1.
Examples:
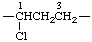 |  |
| 1-chloropropane-1,3-diyl | cyclohexane-1,4-diyl |
In polycyclic hydrocarbons, bridged hydrocarbons, spiro hydrocarbons, ring assemblies, and heterocyclic systems, numbering is fixed for the ring system. The points of attachment of such subunits to the main chain of the CRU should have the lowest permissible locants consistent with the fixed numbering. The same fixed numbering is retained for either direction of progress through the group in generating the subunit name. Where there is a choice, the point of attachment at the left-hand side of the ring as written in the CRU should have the lowest permissible locant.
Examples:
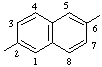 | 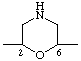 |
| naphthalene-2,6-diyl | morpholine-2,6-diyl |
The factors in the selection of the preferred CRU are in order:
seniority of subunitsIn selecting the preferred CRU, the steps to be followed in sequence are:path length between subunits
| 1. | write the structure of a representative portion of the polymer chain; |
| 2. | identify subunits and substituents; |
| 3. | classify the subunits according to their seniority; |
| 4. | find the shortest path in atoms, irrespective of their nature, from the subunit of the highest seniority to the subunit of the same seniority (Rules 15, 16), if present, or of the second highest seniority (Rules 13, 14). Where paths of equal length are identified as shortest, the choice depends on the seniority of the remaining subunits and the number and positions of substituents; |
| 5. | orient the structure so that the direction from the most senior subunit to the subunit of the next highest seniority reads left to right; |
| 6. | identify the preferred CRU starting from the highest seniority subunit and moving in the direction determined above. |
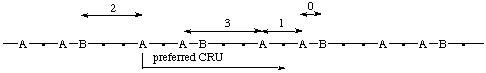
The starting point for the preferred CRU is at the subunit of highest seniority (A). Of the two paths leading from the subunit of highest seniority (A) along the main chain (backbone) to both the next subunits of second highest seniority (B), the shorter path is to be followed.
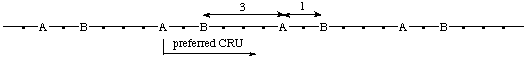
(dots represent subunits of lowest seniority)
(B is closer to A in the preferred CRU, i.e.,  )
)
Example:
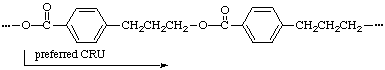
(O is senior to a benzene ring; the one-atom path from O to the benzene ring is preferred to the three-atom path)
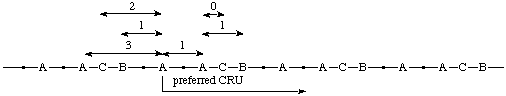
When two paths from the starting subunit (A) to both the next subunits of second highest seniority (B) are equally long, the shorter path from the starting subunit A to the subunit of third highest seniority (C) is to be followed.
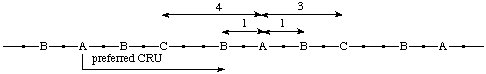
Example:
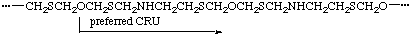
(O is senior to S, which is senior to N; the one-atom path from O to S is to be followed by the six-atom path from S to O in which N is closer to S)
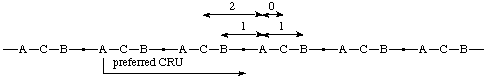
Example:
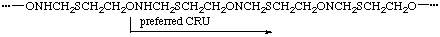
(O is senior to S, which is senior to N; of the two equal paths from O to S, the one that traverses N is preferred to that which traverses CH2)
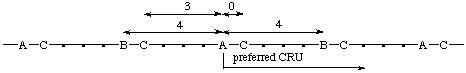
(C is closer to A in the preferred CRU; both B's are equally distant from A)
Example:
(O is senior to N, which is senior to a benzene ring; of the two equal paths from O to N, the one that traverses the benzene ring earlier is preferred)
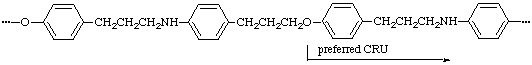
When two identical subunits of the highest seniority (A) are present in a CRU in the main chain (backbone), the shorter path between the identical subunits is to be followed. The starting point is chosen in such a way that the shorter path to the subunit of second highest seniority (B) is followed. If the paths are equal, the paths to subunits of third highest seniority (C) are considered in the sense of Rule 14.
(B is closer to both A subunits in the preferred CRU)
Example:

(O is senior to S; the one-atom path from O to O is preferred to the three-atom path from O to O; S is closer to O in the preferred CRU)
(of the two equal paths from  to B, the one that traverses C is preferred to that which traverses
to B, the one that traverses C is preferred to that which traverses  )
)
Example:
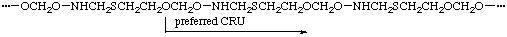
(O is senior to S, which is senior to N; the one-atom path from O to O is preferred to the five-atom path; N is closer to O in the preferred CRU)
When three or more identical subunits of the highest seniority (A) are present in a CRU in the main chain (backbone), the starting point and direction are chosen in such a way that the shortest path through all the subunits A results. If there is a choice, the CRU with the shorter path to the subunits of second or third highest seniority (B or C) is selected.
Examples:
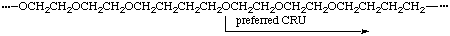
(shortest path through all oxygens, either direction)
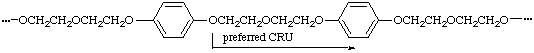
(shortest path through all oxygens)
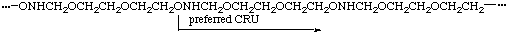
(starting O and direction determined by N)
If a choice is possible between a divalent and a higher-valent CRU, the number of free valences is minimized only after all other orders of seniority have been observed.
Example: