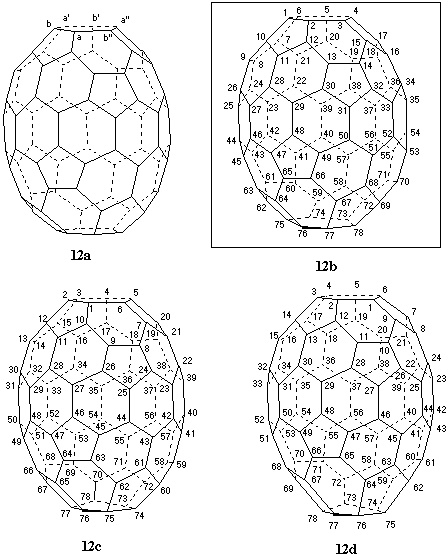
Fig. 12. Systematic numbering of (f,sA)(C78-D3)[5,6]fullerene
This fullerene contains one C3 axis, passing through the center of opposite and symmetry-equivalent six-membered rings (Fig. 12a), and three C2 axes, perpendicular to the C3 axis. According to Fu-3.1.1 the highest order axis (C3) must be examined first for contiguous helical pathways. The atoms of the six-membered ring are symmetry-equivalent in sets of three (a/a'/a" and b/b'/b"). There are four possible pathways to be considered: b to a to b"; a to b to a'; b" to a to b; and a to b" to a". Only the first three (shown in Figs. 12b, 12c, and 12d) lead to contiguous helical numberings terminating at an atom belonging to the six-membered ring intersected by the C3 axis. Rule Fu-3.1.3 does not select among these numberings. Application of Fu-3.1.4 first eliminates the numbering of Fig. 12d, in which there is a 6,6,5 atom at position 22 rather than a 6,6,6 atom as in the numberings of Figs. 12b and 12c. By continuing the atom- ranking exploration for 12b and 12c, the numbering in Fig. 12b is preferred over that in Fig. 12c because it features a 6,6,6 rather than a 6,6,5 atom at position 40. The stereodescriptor for the shown enantiomer of this inherently chiral fullerene is (f,sA) [ref 4].

Fig. 12. Systematic numbering of (f,sA)(C78-D3)[5,6]fullerene
References for this section
3. A.L. Goodson, C.L. Gladys and D.E. Worst, J. Chem. Inf. Comp. Sci. 1995, 35, 969-978.
4. International Union of Pure and Applied Chemistry, Division of Organic Chemistry, Commission on Nomenclature of Organic Chemistry. "Nomenclature for the (C60-Ih) and (C70-D5h(6))[5,6]fullerenes (IUPAC recommendations 2002)", Pure Appl. Chem. 2002, 74, 629-695.
5. P.W. Fowler and D.E. Manolopoulos, An Atlas of Fullerenes, Clarendon Press, Oxford, 1995