Phane Nomenclature, Part II
PhII-6
Polysubstituted derivatives of phane parent hydrides
Continued from PhII-5
Contents of this section
- PhII-6. Polysubstituted derivatives of phane parent hydrides
- PhII-6.1. Principal characteristic groups.
- PhII-6.2. Name component order for polysubstituted phane derivatives.
- PhII-6.3. Order for assignment of lowest locants for phane derivatives.
- References
PhII-6. Polysubstituted derivatives of phane parent hydrides
PhII-6.1. Principal characteristic groups. The order of priority of classes (see Table 10, ref. 3o) is applied to select the principal characteristic group. Characteristic groups that are not selected as the principal characteristic group are cited as prefixes. Numbering of phane parent hydrides is based primarily on the location and nature of their amplificants and on the nature of the simplified skeleton (monocyclic, spiro, bi- and polycyclic). Lowest locants are assigned in conformity with this fixed numbering first to the principal characteristic groups and then to groups cited as prefixes.
Examples:
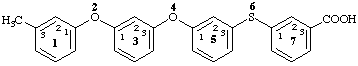
13-methyl-2,4-dioxa-6-thia-1,7(1),3,5(1,3)-tetrabenzenaheptaphane-73-carboxylic acid
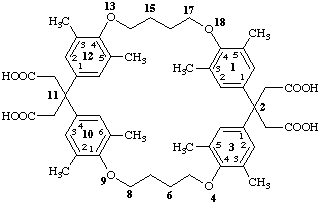
2,2',2'',2'''-[13,15,33,35,102,106,123,125-octamethyl-4,9,13,18-tetraoxa-1,3,10,12(1,4)-tetrabenzenacyclooctadecaphane-2,2,11,11-tetrayl]tetraacetic acid
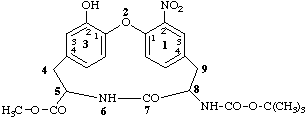
methyl 8-[(tert-butoxycarbonyl)amino]-32-hydroxy-12-nitro-7-oxo-2-oxa-6-aza-1,3(1,4)-dibenzenacyclononaphane-5-carboxylate
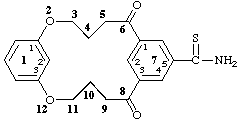
6,8-dioxo-2,12-dioxa-1,7(1,3)dibenzenacyclododecaphane-75-carbothioamide
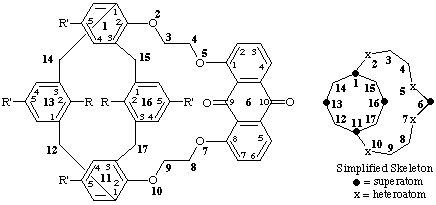
R = -OH ; R' = -C(CH3)3
15,115,135,165-tetra-tert-butyl-132,162-dihydroxy-2,5,7,10-tetraoxa-6(1,8)-anthracena-1,11(2,1,3),13,16(1,3)-tetrabenzenabicyclo[9.3.3]heptadecaphane-69,610-dione
(for the nomenclature of quinones, see ref. 3p)
R = -CH2-COO-CH2-CH2-CH3 R' = -C(CH3)3
dipropyl 2,2'-[15,115,135,165-tetra-tert-butyl-69,610-dioxo-69,610-dihydro-2,5,7,10-tetraoxa-6(1,8)-anthracena-1,11(2,1,3),13,16(1,3)-tetrabenzenabicyclo[9.3.3]heptadecaphane-132,162-diyl]diacetate
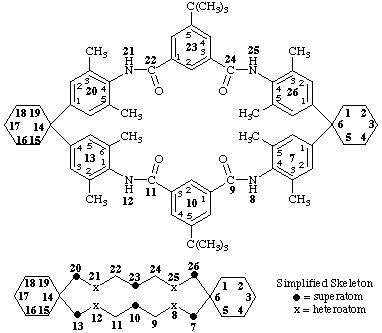
105,235-di-tert-butyl-73,75,132,136,203,205,263,265-octamethyl-8,12,21,25-tetraaza-7,13,20,26(1,4),10,23(1,3)-hexabenzena-
dispiro[5.7.514.76]hexacosaphane-9,11,22,24-tetrone
PhII-6.2. Name component order for polysubstituted phane derivatives.
Writing of polysubstituted phane names follows the normal order for writing names for organic compounds. The name components for polysubstituted phane derivatives are cited in the following order, from left to right:
1. detachable prefixes (alphabetized);
2. nondetachable 'hydro-'/'dehydro-' prefixes;
3. nondetachable replacement 'a' prefixes ;
4. parent hydride name ;
5. '-ene' and '-yne' endings ;
6. suffixes.
Example:
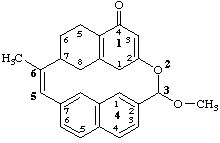
3-methoxy-6-methyl-15,16,17,18-tetrahydro-2-oxa-1,4(2,7)-dinaphthalenacyclohexaphan-5-en-14(11H)-one
(added hydrogen method, see PhII-3.2)
3-methoxy-6-methyl-11,14,15,16,17,18-hexahydro-2-oxa-1,4(2,7)-dinaphthalenacyclohexaphan-5-en-14-one
(nondetachable 'hydro-' prefixes, see PhII-3.2)
PhII-6.3. Order for assignment of lowest locants for phane derivatives.
When necessary, lowest locants are attributed to the following structural features considered successively in the order listed until a decision is reached (see PhII-1.1):
1. principal group named as suffix;
2. unsaturation ('-ene'/'-yne' endings);
3. substituents named as prefixes:
Examples:
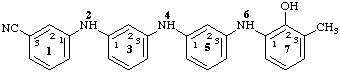
72-hydroxy-73-methyl-2,4,6-triaza-1,7(1),3,5(1,3)-tetrabenzenaheptaphane-13-carbonitrile
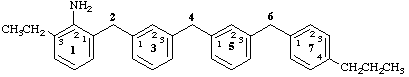
13-ethyl-74-propyl-1,7(1),3,5(1,3)-tetrabenzenaheptaphan-12-amine
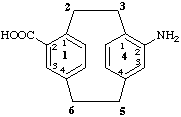
42-amino-1,4(1,4)-dibenzenacyclohexaphane-12-carboxylic acid
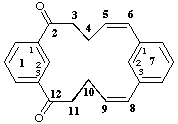
1,7(1,3)-dibenzenacyclododecaphane-5,8-diene-2,12-dione
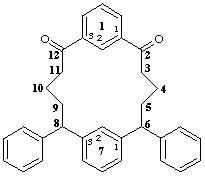
6,8-diphenyl-1,7(1,3)dibenzenacyclododecaphane-2,12-dione
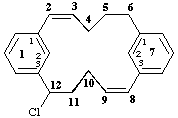
12-chloro-1,7(1,3)-dibenzenacyclododecaphan-2-ene
References
1. International Union of Pure and Applied Chemistry. Organic Chemistry Division. Commission on Nomenclature of Organic Chemistry."Phane Nomenclature, Part I: Phane Parent Names (IUPAC Recommendations 1998)" Pure Appl. Chem. 70, 1513-1545 (1998). (a) PhI-2.2, pp. 1522-1523; (b) PhI-3.3, pp. 1534-1536. (See also IUPAC chemical nomenclature database https://iupac.qmul.ac.uk/phane/).
2. International Union of Pure and Applied Chemistry. Organic Chemistry Division. Commission on Nomenclature of Organic Chemistry/Commission on Physical Organic Chemistry. "Glossary of Class Names of Organic Compounds and Reactive Intermediates Based on Structure" Pure Appl. Chem. 67, 1307-1375 (1995) (See also IUPAC chemical nomenclature database https://iupac.qmul.ac.uk/class/).
3. International Union of Pure and Applied Chemistry. Organic Chemistry Division. Commission on Nomenclature of Organic Chemistry. A Guide to IUPAC Nomenclature of Organic Compounds, Recommendations 1993, R. Panico, W. H. Powell and J.-C. Richer, eds. Blackwell Scientific Publications, Oxford, 1993. (a) R-4.1, pp. 68-72; (b) R-5.7.4.2, pp. 117-119; (c) R-5.7.7, pp. 123-125; (d) R-5.7.6, pp. 122-123; (e) R-4.1, p. 72; (f) R-0.1.8, pp. 10-12; (g) R-0.2.4.2, p. 17; (h) R-1.3, pp. 34-35; (i) R-2.5, pp. 56-58; (j) R-0.1.8.3, pp. 10-12; (k) R-3.1.2, pp. 60-61; (m) R-3.1.3, p. 61; (n) R-3.1.1, pp. 59-60; (o) Table 10, p. 70; (p) R-5.6.2.1, pp. 100-101. (See also http://www.acdlabs.com/iupac/nomenclature/).
4. International Union of Pure and Applied Chemistry. Organic Chemistry Division. Commission on Nomenclature of Organic Chemistry. "Revised Nomenclature for Radicals, Ions, Radical Ions and Related Species (IUPAC Recommendations 1993)" Pure Appl. Chem. 65, 1357-1455 (1993). (a) RC-82.4, pp. 1411-1413. (See also IUPAC chemical nomenclature database https://iupac.qmul.ac.uk/ions/).
5. International Union of Pure and Applied Chemistry and International Union of Biochemistry and Molecular Biology. IUPAC-IUB Joint Commission on Biochemical Nomenclature (JCBN). "Nomenclature of Tetrapyrroles, Recommendations 1986". Eur. J. Biochem. 178, 277-328 (1988), TP-5.4, p. 302; Biochemical Nomenclature and Related Documents, A Compendium, Second Edition, 1992, Portland Press Ltd, London, p. 303. (See also IUPAC chemical nomenclature database https://iupac.qmul.ac.uk/tetrapyrrole/).
6. International Union of Pure and Applied Chemistry. Organic Chemistry Division. Commission on Nomenclature of Organic Chemistry. "Nomenclature of Fused and Bridged Fused Ring Systems (IUPAC Recommendations 1998" Pure Appl. Chem. 70, 143-216, 1998. (See also IUPAC chemical nomenclature database https://iupac.qmul.ac.uk/fusedring/).
Return to Phane nomenclature part II home page
Return to IUPAC nomenclature home page












