- A. Generic terms (Lip-2.1 to Lip-2.10)
- B. Individual compounds (Lip-2.11 to Lip-2.12)
I. FATTY ACID, NEUTRAL FATS, LONG-CHAIN ALCOHOLS AND LONG-CHAIN BASES
A. Generic Terms
Lip-1.1. The term 'fatty acid' designates any one of the aliphatic monocarboxylic acids that can be liberated by hydrolysis from naturally occuring fats and oils. In the terms 'free fatty acids' or 'nonesterified fatty acids', now widely in use, 'free' and 'nonesterified' are actually redundant and should be omitted (See Lip-1.14). [The designation 'aliphatic carboxylate (C10-C26, nonesterified)' used by the Commission on Quantities and Units in Clinical Chemistry is correct, but rather cumbersome.] Whenever the sum of fatty acids and their esters is determined by an analytical method, this should be explicitly stated. (See also Lip-1.6).
Lip-1.2. 'Neutral fats' are mono-, di-, or triesters of glycerol with fatty acids, and are therefore termed monoacylglycerol, diacylglycerol, or triacylglycerol, as appropriate. 'Acylglycerols' includes mixtures of any or all of these.
Comments. (a) The term 'acyl' is used in Organic Nomenclature [3] to denote the radical formed by loss of the OH group from the acid function of any acid (cf. Lip-1.6). We are concerned here with acyl radicals of aliphatic carboxylic acids with four or more carbon atoms, the larger members of which (> C10) are also known as 'higher fatty acids'.
(b) The old terms monoglyceride, diglyceride, and triglyceride are discouraged and should progressively be abandoned, not only for consistency, but mainly because strict interpretation does not convey the intended meaning. 'Triglyceride', taken literally, indicates three glycerol residues (e.g., cardiolipin), diglyceride two (e.g., phosphatidyIglycerol), and a monoglyceride is a monoacy1glycerol.
Lip-1.3. The generic term 'long-chain alcohol' or 'fatty alcohol' refers to an aliphatic compound with a chain-length greater than C10 that possesses a terminal CH2OH group. Such alcohols should be named according to systematic nomenclature [3]. (See Lip-1.7).
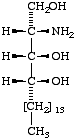
Lip-1.4. The term 'sphingoid' or 'sphingoid base' refers to sphinganine (cf. Lip-1.8), [D-erythro-2-amino-1,3-octadecanediol (I)], to its homologs and stereoisomers (II, III), and to the hydroxy and unsaturated derivatives of these compounds (IV-VI). The term 'long-chain base' may be used in a wider sense to indicate any base containing a long-chain aliphatic radical.
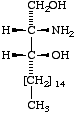 | 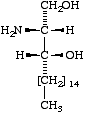 | 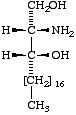 |
| I Sphinganine [D-erythro or 2S,3R configuration implied] | II (2R,3R)- (or D-threo-)-2-Amino- 1,3-octadecanediol | III Icosasphinganine (formerly eicosasphinganine, see footnoted in Appendix A) |
 | 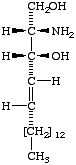 | 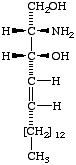 |
| IV (2S,3S,4R)-2-amino- 1,3,4-octadecanetriol; (phytosphingosine). | V Sphingosine; (4E)-sphingenine; trans-4-sphingenine; (2S,3R,4E)-2-amino-4-octadecene-1,3-diol; | VI cis-4-Sphingenine; (4Z)-sphingenine. |
(a) sphingolipid, for any lipid containing a sphingoid;
(b) ceramide, for an N-acylated sphingoid;
(c) sphingomyelin, for a ceramide-1-phosphocholine. (See [2] for this use of 'phospho'; also Lip-2.11);
(d) glycosphingolipid, for any lipid containing a sphingoid and one or more sugars. (See below for other generic terms).
B. Individual Compounds
1. Fatty Acids and Alcohols
Lip-1.6. Fatty acids (cf. Lip-1.1) and their acyl radicals (cf. Lip-1.2, comment [a]) are named according to the IUPAC Rules for the Nomenclature of Organic Chemistry ([3], Rule C-4). (A list of trivial names is given in Appendix A.) Fatty acids are numbered with the carbon atom of the carboxyl group as C-1. By standard biochemical convention, the ending '-ate' in, e.g., palmitate denotes any mixture of the free acid and its ionized form in which the cations are not specified. The ending '-ate' is also used to designate esters, e.g., cholesteryl palmitate, ethylidene dilaurate, etc. (cf. Lip-1.12). Structural isomers of polyunsaturated acids, hitherto distinguished by Greek letters (e.g., a- and g-linolenic acids), are better distinguished by the locants of the unsaturated linkages [e.g. (9,12,15)- and (6,9,12)-linolenic acids, respectively]. (See Lip-1.15). However, the Greek letter prefixes may be useful in (defined) abbreviations (see Appendix A).
Lip-1.7. Long-chain alcohols (fatty alcohols) and the radicals derived from them should be designated by their systematic names ([3], Rules C-201 and A-1 et seq.), but not by trivial names that are derived from those of fatty acids.
Examples:
(a) 1-hexadecanol and hexadecyl-, not palmityl alcohol and palmityl-;
(b) 1-dodecanol and dodecyl-, not lauryl alcohol and lauryl-.
2. Sphinganine and Derivatives
Lip-1.8. The compound previously known as dihydrosphingosine [D-erythro-2-amino-1,3-octadecanediol or (2S,3R)-2-amino-1,3-octadecanediol] is called sphinganine (1).
Lip-1.9. Trivial names of higher or lower homologs of sphinganine may be derived by adding a prefix ([3], Rule A-1) denoting the total number of carbon atoms in the main chain of the homolog, e.g., icosasphinganine for the C20 compound (III), hexadecasphinganine for the C16 compound.
Note. See footnote d in Appendix A re 'icosa' for 'eicosa'.
Lip-1.10. The recommendations on the Nomenclature of Glycolipids imply that parts of Lip-1.10 are no longer recommended. The following version has a text edited to reflect current recommendations. Readers who wish to consult the original wording can see this as an appendix.
Affixes denoting substitution of sphinganine (hydroxy, oxo, methyl, etc.) are used as usual, according to existing rules [3]. The configuration is indicated using the R/S system [6].
Sphingoids differing from sphinganine in their configurations at C-2 and/or C-3 should be named not as derivatives of sphinganine, but with fully systematic names [3], using the prefixes D-threo, L-erythro, as appropriate, e.g., D-threo-2-amino-1,3-octadecanediol, or (2R,3R)-2-amino-1,3-octadecanediol, for II (cf. Rule Carb-8 in [5]). (Cf. Lip-1.11, example [d]).
Comments. (a) The semisystematic names for the sphingoids are significantly shorter than the fully systematic names only if the terms chosen imply not only substituents but also configurations. Therefore, the name 'sphinganine' specifies the D-erythro configuration, and it is logical that the names of stereoisomers of sphinganine differing in configuration at C-2 and/or C-3 should not include 'sphinganine' as a root. This recommendation differs from that in the previous document [1].
(b) The configurational prefixes using the R/S system [6] may change with changes in the substitution. For example C-3 is R in icosasphinganine (III) but S in 4-hydroxysphinganine (IV).
Examples:
(a) (2R,3R)-2-amino-1,3-octadecanediol, for II;
(b) (2S,3S,4R)-2-amino-1,3,4-octadecanetriol for IV;
(c) (2S,3R,4E)-2-amino-4-octadecene-1,3-diol for sphingosine (V) (See also Lip-1.11).
Lip-1.11. Names for unsaturated compounds are derived from the names of the corresponding saturated compounds by the appropriate infixes, namely ene, diene, yne, etc. [3]. If the geometry of the double bond is known, it should be indicated by the more modern E-Z system (cf. [6], Rule E-2.2), e.g., (4E)-sphingenine for sphingosine (V).
Comment. The trivial name 'sphingosine' (V) is retained. If trivial names other than sphingosine are used, they should be defined in each paper in terms of this nomenclature, or of the general nomenclature of organic chemistry [3].
Other names for compounds described in Lip-1.10 and Lip-1.11 :
(a) omitted, see appendix;
(b) (4E)-sphingenine for sphingosine (V);
(c) (4Z)-sphingenine for the geometric isomer of sphingosine (VI);
(d) D-threo-2-amino-1,3-octadecanediol for the C-2 epimer of sphinganine (II); cf. Lip-1.10, example (a).
3. Glycerol Derivatives
Lip-1.12. Esters, ethers and other O-derivatives of glycerol are designated according to Carb-15 of the Rules of Carbohydrate Nomenclature [5], i.e. by a prefix, denoting the substituent, preceded by a locant. if the substitution is on a carbon atom, the compound is designated by its systematic name and not as a derivative of glycerol (e.g., 1,2,3-nonadecanetriol for C16H33CHOH-CHOH-CH2OH, which could be considered as 1-C-hexadecy1glycerol). It is permissible to omit the locant 'O' if the substitution is on the oxygen atoms of glycerol.
Examples:
(a) tristearoylglycerol or tri-O-stearoyl glycerol or glycerol tristearate, or glyceryl tristearate;
(b) 1,3-benzylideneglycerol or 1,3-O-benzylideneglycerol;
(c) glycerol 2-phosphate (a permissible alternative to this term is 2-phosphoglycerol) [10].
Comment. The alternative system set forth in Carb-16 of the Rules on Carbohydrate Nomenclature [5], i.e. the use of the suffix '-ate', is less suitable for glycerol esters, with the exception of the phosphates (see Examples). However, this system may be used to designate esters of monofunctional alcohols, e.g. cholesteryl palmitate (cf. Lip-1.6).
Lip-1.13. Stereospecific Numbering. In order to designate the configuration of glycerol derivatives, the carbon atoms of glycerol are numbered stereospecifically. The carbon atom that appears on top in that Fischer projection that shows a vertical carbon chain with the hydroxyl group at carbon-2 to the left is designated as C-1. To differentiate such numbering from conventional numbering conveying no steric information, the prefix 'sn' (for stereospecifically numbered) is used. This term is printed in lower-case italics, even at the beginning of a sentence, immediately preceding the glycerol term, from which it is separated by hyphen. The prefix 'rac-' (for racemo) precedes the full name if the product is an equal mixture of both antipodes; the prefix 'X-' may be used when the configuration of the compound is either unknown or unspecified (cf. Lip-1.10).
Examples:
(a) sn-glycerol 3-phosphate for the stereoisomer (VII = VIII), previously known as either L-a-glycerophosphate or as D-glycerol 1-phosphate;
(b) rac-1-hexadecylglycerol;
(c) 1,2-dipalmitoyl-3-stearoyl-X-glycerol.
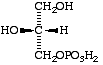 |  | |
| VII | VIII | |
| sn-Glycerol 3-phosphate | ||
| L-(glycerol 3-phosphate) | D-(glycerol 1-phosphate) |
Comments. (a) The problem of distinguishing between stereoisomers was discussed in extenso in the 1967 document [1]. In brief, difficulties arise because glycerol is a prochiral compound. The parent substance of many phospholipids, natural glycerol phosphate, has been named both as L-a-glycerol phosphate [7] (VII) and, according to standard rules of nomenclature, D-glycerol 1-phosphate [8] (VIII). When the R/S system (sequence rule) is applied, substitution of one of the primary hydroxyl groups often leads to changes in the configurational prefix, thus obscuring chemical and biogenetical relationships; it is generally inapplicable to the steric description of such mixtures as occur in triacy1glycerols isolated from natural sources. The stereospecific numbering of glycerol and its derivatives as proposed by Hirschmann [9], described above and in [1], avoids these difficulties; it has proved useful and is widely accepted.
(b) The enantiomer of sn-glycerol 3-phosphate (VII) is sn-glycerol 1-phosphate (IX), as is evident from the structures.
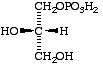
IX
sn-Glycerol 1-phosphate [L-(glycerol 1-phosphate) D-(glycerol 3-phosphate)]
C. Symbols and Abbreviations
Lip-1.14. The term 'fatty acids' (cf. Lip-1.1) should not be abbreviated. The use of abbreviations like 'FFA' for 'free fatty acids' or 'NEFA' for 'non-esterified fatty acids' is strongly discouraged.
Comment. The words 'acids' and 'esters' serve to distinguish the 'free' (nonesterified) and 'bound' (esterified) fatty acids and are as short or shorter than the abbreviations themselves.
Lip-1.15. In tables and discussions where various fatty acids are involved, the notation giving the number of carbon atoms and of double bonds (separated by a colon) is acceptable, e.g. 16:0 for palmitic acid, 18:1 for oleic acid. Also, terms such as '(18:0)acyl' may be used to symbolize radicals of fatty acids. (See Appendix A).
Comment. This system is already widely used. It should, however, be kept in mind that it sometimes does not completely specify the fatty acid. For example, a-linolenic acid and g-linolenic acid are both 18:3 acids; the designation 18:3 is therefore ambiguous. In such a case, the position of double bonds sholild be indicated, e.g. 18:3(9,12,15) for (9,12,15)-linolenic acid, formerly known as a-linolenic acid.
Lip-1.16. It is sometimes desirable (for example, in discussing the biosynthesis of lipids) to indicate the position of each double bond with reference not to the carboxyl group (always C-1), but to the end of the chain remote from the carboxyl. If n is the number of carbon atoms in the chain (i.e. the locant of the terminal methyl group) and x is the (lower) locant of the double bond, the position of the double bond may be defined as (n minus x). Thus, the common position of the double bond in oleic and nervonic acids may be given as 18-9 and 24-9, respectively. This structural regularity should not be expressed as w9.
Lip-1.17. The system described in Lip-1.15 may also be used to denote alcohols and aldehydes related to fatty acids, provided that the nature of the residue is clearly indicated either by the appropriate name of the compound(s) (e.g. 18:1 alcohol) or in the heading of the table. The 1-ene of alk-1-en-1-yl (i.e. 1-alkenyl) compounds is not counted in this system (see Lip-2.10, comment).
Lip-1.18. For many complex lipids, a representation of the structures using symbols rather than structural formulae may be useful. Symbols proposed for various constituents are given in Appendix B (see also [10]), and, for glycolipids, in Lip-3.13. They are constructed in analogy to those in use for amino acids [11], nucleosides [12] and saccharides [13].
II. PHOSPHOLIPIDS
A. Generic Terms
The Rules of the Nomenclature of Organic Phosphorus Compounds, also known as D-Rules [2] recognize, for biochemical usage, the prefix 'phospho' as an alternate to 'O-phosphono-' (or 'N-phosphono-'). By a similar convention [10], '-phospho-' may be used as an infix to designate the phosphodiester bridge present in such compounds as glycerophosphocholine. The use of root names like 'phosphatidic acid' is retained and extended (Lip-2.3 to Lip-2.4).
Lip-2.1. 'Phospholipid' may be used for any lipid containing phosphoric acid as mono- or diester. Likewise, lipids containing C-phosphono groups (e.g. compound X) may be termed 'phosphonolipids'.
Lip-2.2. 'Glycerophospholipid' signifies any derivative of glycerophosphoric acid that contains at least one O-acyl, or O-alky], or O-(1-alkenyl) group attached to the glycerol residue. Generic names for other classes of phospholipids may be coined according to this scheme, e.g., sphingophospholipid, inositolphospholipid.
Comment. The old terms, 'phosphatide', 'phosphoglyceride', and 'phosphoinositide' are no longer recommended because they do not convey the intended meaning (see also Lip-1.2).
Lip-2.3. 'Phosphatidic acid' signifies a derivative of a glycerol phosphate (glycerophosphate) in which both remaining hydroxyl groups of glycerol are esterified with fatty acids. The position of the phosphate group may be emphasized by stereospecific numbering.
Comment. For the most common (3-sn) phosphatidic acid and its derivatives, the locants are often omitted. However, 'phosphatidyl' without locants can lead to ambiguities. It is therefore preferable to use the proper locants, for example, 2-phosphatidic acid for compound XI, and 3-sn-phosphatidylserine for XIIa.
[See also entry from 1980 Newsletter]
Lip-2.4. The common glycerophospholipids are named as derivatives of phosphatidic acid, e.g. 3-sn-phosphatidylcholine (this term is preferred to the trivial name, lecithin; the systematic name is 1,2-diacyl-sn-glycero-3-phosphocholine); 3-sn-phosphatidylserine; 1-phosphatidylinositol [see comment (b) below]; 1,3-bis(3-sn-phosphatidyl)glycerol.
Comments. (a) It is understood that, in combination with compounds like ethanolamine (properly, 2-aminoethanol) or serine, which bear both hydroxyl and amino groups, substitution by phosphorus is at the hydroxyl group of the ethanolamine or serine. Substitution at any other position, or where confusion may arise, requires a locant.
(b) The phosphorylated derivatives of 1-(3-sn-phosphatidyl)inositol should be called 1-phosphatidylinositol 4-phosphate and 1-phosphatidylinositol 3,4-bisphosphate, respectively. The use of 'diphosphoinositide' and 'triphosphoinositide' for these is discouraged, as these names do not convey the chemical structures of the compounds and can be misleading. (Cf. also Table 4 in [10])
Lip-2.5. As an alternative, generic names may be coined according to Lip-1.13, i.e. using glycerol phosphate (glycerophosphate) as the stem. In this case, the stereospecific numbering of glycerol should be used to indicate the position of the phosphoric residue as well as the other substituents (acyl-, alkyl-, 1-alkenyl). If the nature of these substituents cannot be specified, the prefix 'radyl' may be used.
Lip-2.6. Derivatives of phosphatidic acids resulting from hydrolytic removal of one of the two acyl groups may be designated by the old prefix 'lyso', e.g. lysophosphatidylethanolamine for compound XIIb. A locant may be added to designate the site of (hydro)lysis, 2-lyso designating hydrolysis at position 2, leaving a free hydroxyl group at this carbon atom.
Comment. The 'lyso' term originated from the fact that these compounds are hemolytic. It is here redefined to indicate a limited hydrolysis of the phosphatidyl derivative (i.e. 'deacyl').
Lip-2.7. The term 'plasmalogen' may be used as a generic term for glycerophospholipids in which the glycerol moiety bears an 1-alkenyl ether group.
Lip-2.8. The term 'plasmenic acid' signifies a derivative of sn-glycero-3-phosphate in which carbon-1 bears an O-(l-alkenyl) residue, and position 2 is esterified with a fatty acid (XIII). This term can also be used to name derivatives, e.g. plasmenylethanolamine.
Comments. (a) The use of 'phosphatidyl' as a name for the acyl radical of phosphatidic acid has facilitated the nomenclature of its various compounds (see Lip-2.4). Therefore, it seems logical to offer a similar short term for XIII, i.e. 'plasmenic acid', as an alternative to the more systematic name, 2-acyl-1-alkenyl-sn-glycerol 3-phosphate, which, of course, may be used if desired. 'Plasmenic' is a contraction of 'plasmalogenic' and may be especially useful in naming derivatives, e.g., plasmenylserine.
(b) Isomers like those bearing the phosphate residue in position 2 (e.g. compound XIV) should not be named in this way but as derivatives of the corresponding glycerophosphate, using stereospecific numbering.
Lip-2.9. The term 'lysoplasmenic acid' may be used for a derivative of sn-glycero-3-phosphate that has an O-(1-alkenyl) residue on carbon-1, the hydroxyl group in position 2 being unsubstituted (XVa). This name may also be used in combinations like 'lysoplasmenylethanolamine' (XV b).
Lip-2.10. For compounds of type XVI, bearing a saturated ether group in position 1 and an acyl group in position 2 of sn-glycero-3-phosphate, the term 'plasmanic acid' is proposed. Compounds deacylated in position 2, or with a substituent on the phosphoric residue, can be treated as are the plasmenic acids (Lip-2.9).
Comment. The proposed names will be especially useful for naming phosphoric diesters (phosphodiesters), e.g., plasmanylethanolamine, instead of 2-acyl-1-alkyl-sn-glycero-3-phosphoethanolamine. The terms 'plasmanic acid' and 'plasmanyl' may also be applied to ethers with an alkyl group bearing a double bond within the chain, e.g. a 9-hexadecenyl residue (derived from palmitoleic acid). In such cases, the proper term 'alkenyl', if used without the 'ene' locant(s), would be misleading. (See Lip-1.17.)
B. Individual Compounds
Lip-2. 11. Individual glycerophospholipids in which the substituents can be specified are named according to existing Rules [2, 3, 5, 6], using the infix '-phospho-' [2, 10] to indicate the phosphodiester bridge.
Example: 1-palmitoyl-2-stearoyl-sn-glycero-3-phosphoethanolamine for compound XVII.
Lip-2.12. The ketone derived from glycerol, 1,3-dihydroxy-2-propanone, also known as dihydroxyacetone, may be termed 'glycerone', if desired. The name is a contraction of 'glyceroketone' and may be useful to emphasize the relationship with glycerol, glyceraldehyde (glyceral), and glycerate. It also permits a simple symbolism (Appendix B) and the naming of derived lipids, e.g., 1-palmitoyl-3-phosphoglycerone.
1. IUPAC-IUB Commission on Biochemical Nomenclature (1967) Eur. J. Biochem. 2, 127-131, also 12, 1 (1970); [see also Biochemistry, 6, 3287-3292 (1967); Biochem. J., 105, 897-902 (1967); J. Biol. Chem., 242, 4845-4849 (1967); Hoppe-Seyler's Z. Physiol. Chem., 350, 279-285 (1969) (in German)].
2. International Union of Pure and Applied Chemistry (1973) Information Bulletin, Appendix 31. [See also Nomenclature of Organic Chemistry, Sections A, B, C, D, E, F, and H, Pergamon Press, 1979. Edited by J Rigaudy and S P Klesney.]
3. International Union of Biochemistry (1966) Nomenclature of Organic Chemistry (Sections A, B and C) 2nd edn, Butterworths, London. [See also Nomenclature of Organic Chemistry, Sections A, B, C, D, E, F, and H, Pergamon Press, 1979. Edited by J Rigaudy and S P Klesney.]
4. Mills, J. A. & Klyne, W. (1954) Progr. Stereochem. 1, 181.
5. IUPAC Commission on the Nomenclature of Organic Chemistry and IUPAC-IUB Commission on Biochemical Nomenclature (1971) Eur. J. Biochem. 21, 455-477, also 25, 4 (1972) [now revised as Nomenclature of Carbohydrates (1996) Carb-15 is now 2-Carb-24.1].
6. International Union of Pure and Applied Chemistry (1970) J. Org. Chem. 35, 2849-2867; also Eur. J. Biochem. 18, 151-170 (1971) [now revised as Nomenclature of Organic Chemistry: Section E].
7. Baer, E. & Fischer, H. O. L. (1939) J. Biol. Chem. 128, 475.
8. Baddiley, L, Buchanan, J. G. & Carss, B. (1957) J. Chem. Soc. 1869.
9. Hirschmann, H. (1960) J. Biol. Chem. 235, 2762.
10. IUPAC-IUB Commission on Biochemical Nomenclature (1977) Proc. Natl. Acad. Sci. U.S.A. 74, 2222-2230; Hoppe-Seyler's Z. Physiol. Chem. 358, 599-616 (1977); also Eur. J. Biochem. 79, 1-11 (1977) [see also Biochem. J., 1978, 171, 1-19; Biochemical Nomenclature and Related Documents, 2nd edition, Portland Press, 1992, pages 256-264 (Nomenclature of phosphorus-containing compounds of biochemical importance)].
11. IUPAC-IUB Commission on Biochemical Nomenclature (1972) Eur. J . Biochem. 27, 201-207 [also in Biochem. J., 126, 92-97 (1972); Biochim. Biophys. Acta, 263, 205-212 (1972); J. Biol. Chem., 247, 977-983 (1972); Rev. Soc. Quim. Mex., 19, 33-38 (1975); now revised and incorporated in Nomenclature and Symbolism for Amino Acids and Peptides (1983)].
12. IUPAC-IUB Commission on Biochemical Nomenclature (1970) Eur. J. Biochem. 15, 203-208 [see also Arch. Biochem. Biophys. 1971, 145, 425-436; Biochem. J., 1971, 120, 449-454; Biochemistry, 1971, 9, 4022-4027; Biochim. Biophys. Acta 1971, 247, 1-12; 1972, 25, 1; J. Biol. Chem., 1970, 245, 5171-5176; J. Mol. Biol., 1971, 55, 299-310; Pure Appl. Chem., 1974, 40, 277-290; Biochemical Nomenclature and Related Documents, 2nd edition, Portland Press, 1992, pages 109-114].
13. IUPAC-IUB Combined Commission on Biochemical Nomenclature (1967) Eur. J. Biochem. 1, 259-266 [see also Bull. Soc. Chim. Biol. 50, 3-20 (1968) (in French)].
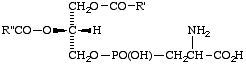
X
A phosphonolipid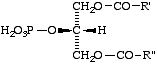
XI
2-Phosphatidic acid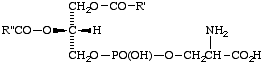
XIIa
Phosphatidylserine
(3-sn-phosphatidylserine)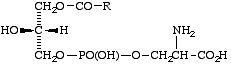
XIIb
A 2-lysophospholipid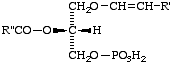
XIII
A plasmenic acid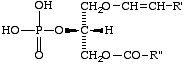
XIV
3-Acyl-1-(1-alkenyl)-sn-glycerol 2-phosphate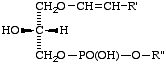
XVa: (R" = H): a lysoplasmenic acid
XVb: (R" = CH2CH2NH2) a lysoplasmenylethanolamine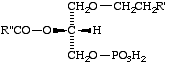
XVI
A plasmanic acid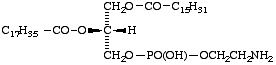
XVII
1-Palmitoyl-2-stearoyl-sn-glycero-3-phosphoethanolamine
References for this Section
Continued with Recommendations Lip-3 and Lip-4
Return to main IUPAC nomenclature home page
Return to main IUBMB nomenclature home page
Return to Biochemical Nomenclature Committees home page