Contents of this section
Parent structures that are not simple derivatives of accepted fundamental parents, but may be considered to arise from such parents by migration of one or more bonds, may be named by the following methods.
RF-4.5.1. The italic prefix x(yz)-abeo- designates the migration of one end of a single bond from its original position in a fundamental parent structure to another position. In this prefix, x is the locant of the stationary, i.e., unchanged, end of the migrating bond; y is the locant of the position of the moving end of the migrating bond in the fundamental parent structure; and z is the locant of the position of the moving end of the migrating bond in the final structure. The numbering of the fundamental parent structure is retained in the new structure.
Note: Previously abeo was italicized (ref 1, 2, 3, 4). For consistency with the other modifying prefixes it is now recommended that a roman font be used. This change was not followed in the lignans and neolignans document (ref 8). The abeo nomenclature described in this recommendation is permissive, not compulsory. It is most suitable for use in discussions of reaction mechanisms and biogenesis. In general systematic names, or names assigned by the homo- and/or nor- method, are preferred.Stereochemical configurations of the fundamental parent structure are retained. New stereochemistry of ring atoms having one hydrogen atom still present is indicated by the α/β system, or, if necessary by the Sequence Rule method (R/S). The projection of the hydrogen atom below (α) or above (β) the reference plane of the ring system is indicated by the appropriate symbol and a capital italic letter H following the locant of the ring atom in the structure, all enclosed in parentheses, and cited before the "abeo-" prefix described above.
Other new stereochemistry is described by the Sequence Rule System.
Examples:
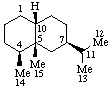 | 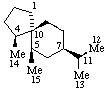 |
| Eremophilane (fundamental parent structure) | (10R)-4(5 |
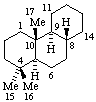 | 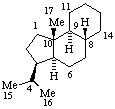 |
| Podocarpane (fundamental parent structure) | (3αH)-5(4 |
Note: The prefix "A-Neo-" has been used to describe the bond migration shown in this last example. However, the "abeo" operation is preferred because it is general.RF-4.5.2. The italic prefix "retro-", preceded by a pair of locants is used to indicate a shift, by one position, of all single and double bonds of a conjugated polyene system delineated by the pair of locants that are not part of a system of maximum number of noncumulative double bonds in a ring or ring system. The first locant is the skeletal atom that has lost a hydrogen atom and the second the one that has gained a hydrogen atom. The prefix retro is used in this manner only in the nomenclature of carotenoids (ref 5).
Note 1: The prefix "retro-" has been used in steroid nomenclature to indicate a 9β,10α-configuration instead of the implied configuration, but is not recommended.Example:Note 2: The prefix "retro-" is also used to designate a peptide in which the amino acid sequence is the reverse of the sequence of the naturally occurring peptide.
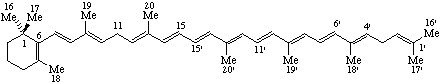
The removal of a terminal ring from a fundamental parent structure of a natural product with the addition of an appropriate number of hydrogen atoms at each junction with the adjacent ring is indicated by the prefix "des-" followed by the capital italic letter of the ring removed (see RF-3.6). Stereochemistry implied by the name of the fundamental parent structure remains the same unless otherwise specified. Numbering of skeletal atoms in the parent structure is retained in the modified structure. This use of 'des-' is restricted to steroids, 3S-7.2. (ref 4).
Examples:
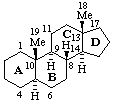 | 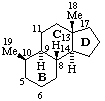 |
| 5α-Androstane (fundamental parent structure) | Des-A-androstane |
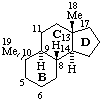
Des-A-10α-androstane
(see RF-10.2.1 for use of α at position 10)
1. International Union of Pure and Applied Chemistry, "Nomenclature of Organic Chemistry. Section F: Natural Products and Related Compounds, Recommendations 1976", IUPAC Information Bulletin Appendices on Tentative Nomenclature, Symbols, Units, and Standards, No. 53, December, 1976. [also in: Eur. J. Biochem. 86, 1-8 (1978)].
2. International Union of Pure and Applied Chemistry, Nomenclature of Organic Chemistry, Sections A, B, C, D, E, F and H, l979 edition, Pergamon Press, Oxford, 1979.
3. International Union of Pure and Applied Chemistry, A Guide to IUPAC Nomenclature of Organic Compounds, Blackwell Scientific Publications, Oxford, 1993. [Corrections see Pure Appl. Chem., 71, 1327-1330 (1999).]
4. International Union of Pure and Applied Chemistry and International Union of Biochemistry, Joint Commission on Biochemical Nomenclature, "Nomenclature of Steroids, Recommendations 1989", Pure Appl. Chem. 61, 1783-1822 (1989). [also in: Eur. J. Biochem. 186, 429-458 (1989) and pages xxx-lix in Dictionary of Steroids (Hill, R.A., Kirk, D.N., Makin, H.L.J. & Murphy, G.M., eds) Chapman & Hall, London 1991].
5. International Union of Pure and Applied Chemistry and International Union of Biochemistry, Commission on Biochemical Nomenclature, "Nomenclature of Carotenoids", Pure Appl. Chem., 41, 405-431 (1975).
8. International Union of Pure and Applied Chemistry, "Nomenclature of Lignans and Neolignans (IUPAC Recommendations 2000)", Pure Appl. Chem., 72, 1493-1523 (2000).